Using Bohr's formula for energy quantization, the ionisation potential of first excited state of hydrogen atom is: . (1) 13.6V, (2) 3.4V, (3) 2.6V, (4) 1.51V

Energy of electron-nth Bohr's orbit-Quick formula - solved problems-IIT JEE NEET-AdiChemistry - YouTube

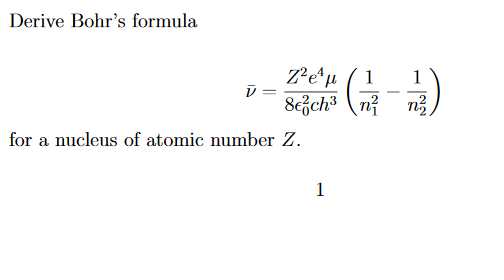

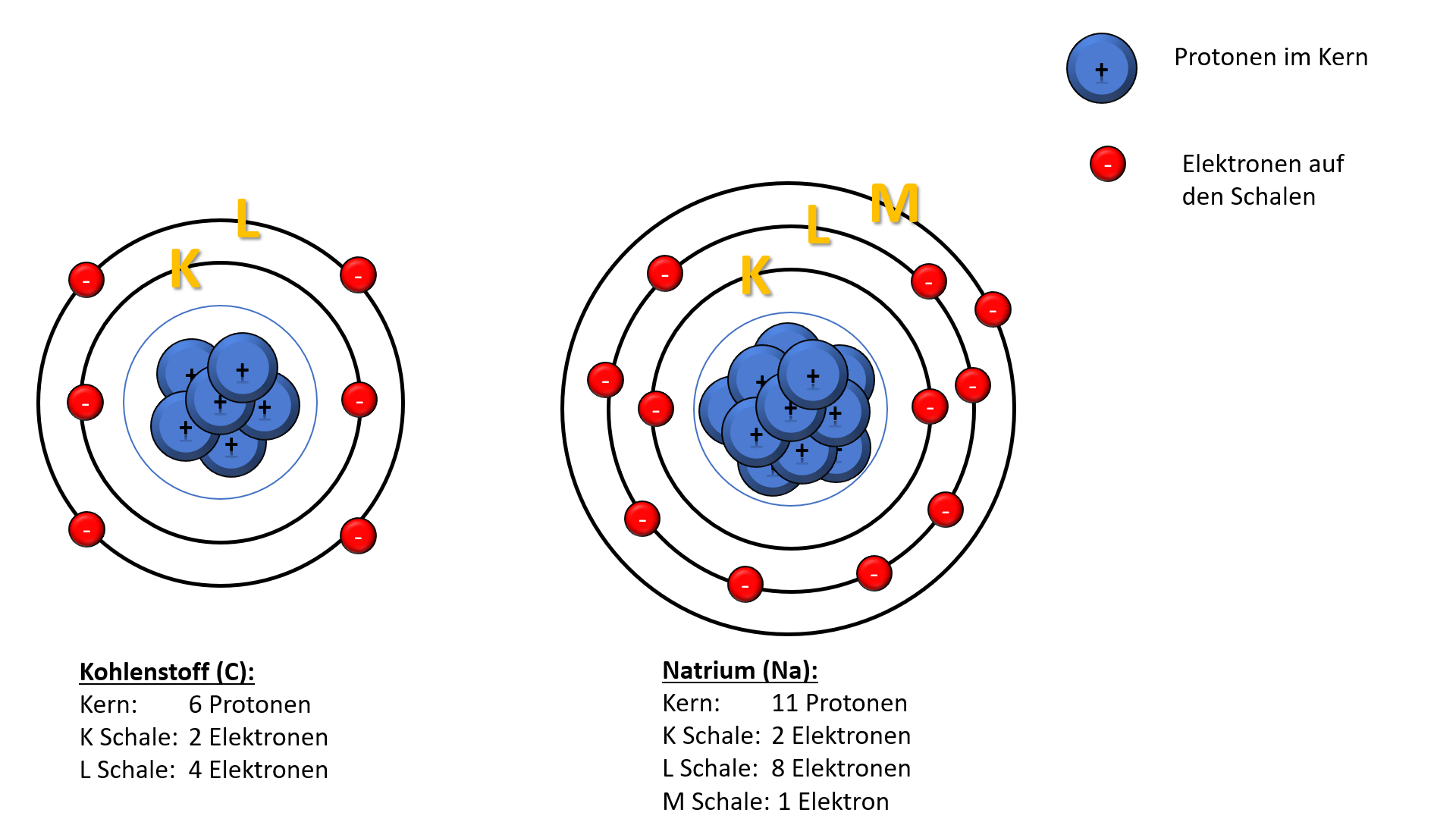
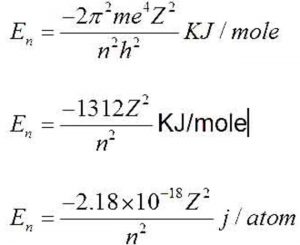
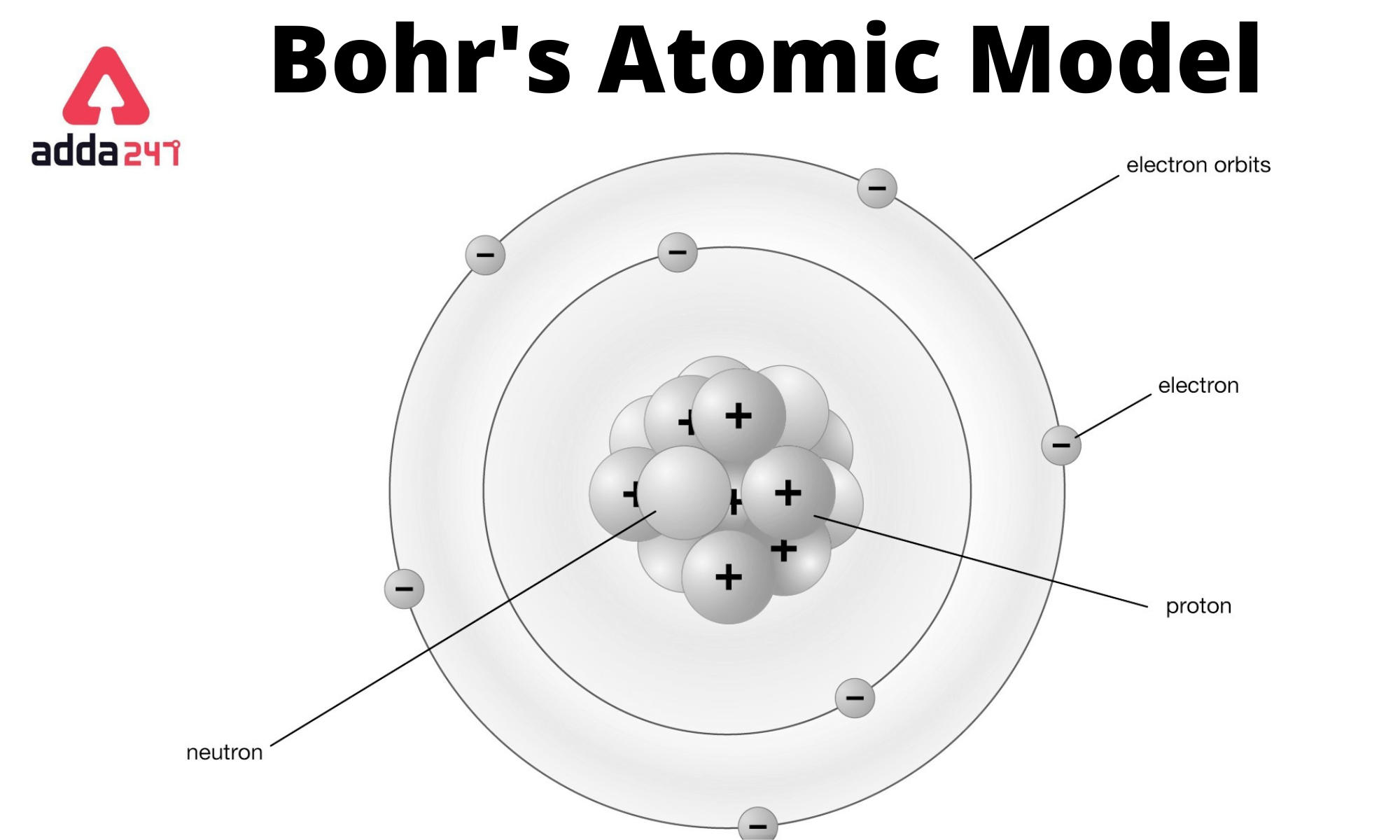



![Bohrsches Atommodell • Erklärung, Postulate, Formulierung · [mit Video] Bohrsches Atommodell • Erklärung, Postulate, Formulierung · [mit Video]](https://d3f6gjnauy613m.cloudfront.net/system/production/videos/001/833/2d5f6fe9ffd6a0af5733f881b21a0a9d4ccb0609/Bohrsches_Atommodell_Thumbnail.png?1628090256)









